Medical Device Solutions
Software Tools and Services Supporting FDA Regulations and Guidance
Services & Automation Software for Medical Device Cybersecurity
Testing
Automation testing platform for FDA expected testing including robustness, fuzz, and penetration testing
Detection
AI/Machine-learning based Intrusion Detection and Protection Software (IDPS) to detect cyber-attacks
Why the FDA is Mandating Security
Several trends are impacting connected medical devices. Cybersecurity is critical as there are financial, data, and operational risks. More importantly, patient safety depends on it.
Security Trends Impacting Healthcare Devices:
- Significant growth in network-connected medical devices – 22% annual growth over the next 5 years.
- Software complexity - more lines if code means more bugs. More bugs means more vulnerabilities.
- Software supply chain is being exploited
- Widely adopted technology, such as Ethernet & Linux, have known exploits
- Skill gap – over 300K unfilled cybersecurity positions

In March 2023 the FDA issued : “Cybersecurity in Medical Devices: Refuse to Accept Policy for Cyber Devices and Related Systems"

A medical device is classified as a “cyber device”, meaning that it needs security, if it:
- Has software
- Has a network interface (or a USB interface)
- Could be adversely affected by a cyber-attack
For pre-market submissions , medical devices manufacturers must:
- Have a process that considers security from the start
- Make sure devices are secure, including testing
- Provide a software bill of materials
- Submit a plan to monitor and address post market cybersecurity vulnerabilities.
- Support a software update process for in field devices to address identified vulnerabilities.
White Paper - Practical Advice for FDA Submissions
Implementing Security for Medical Devices
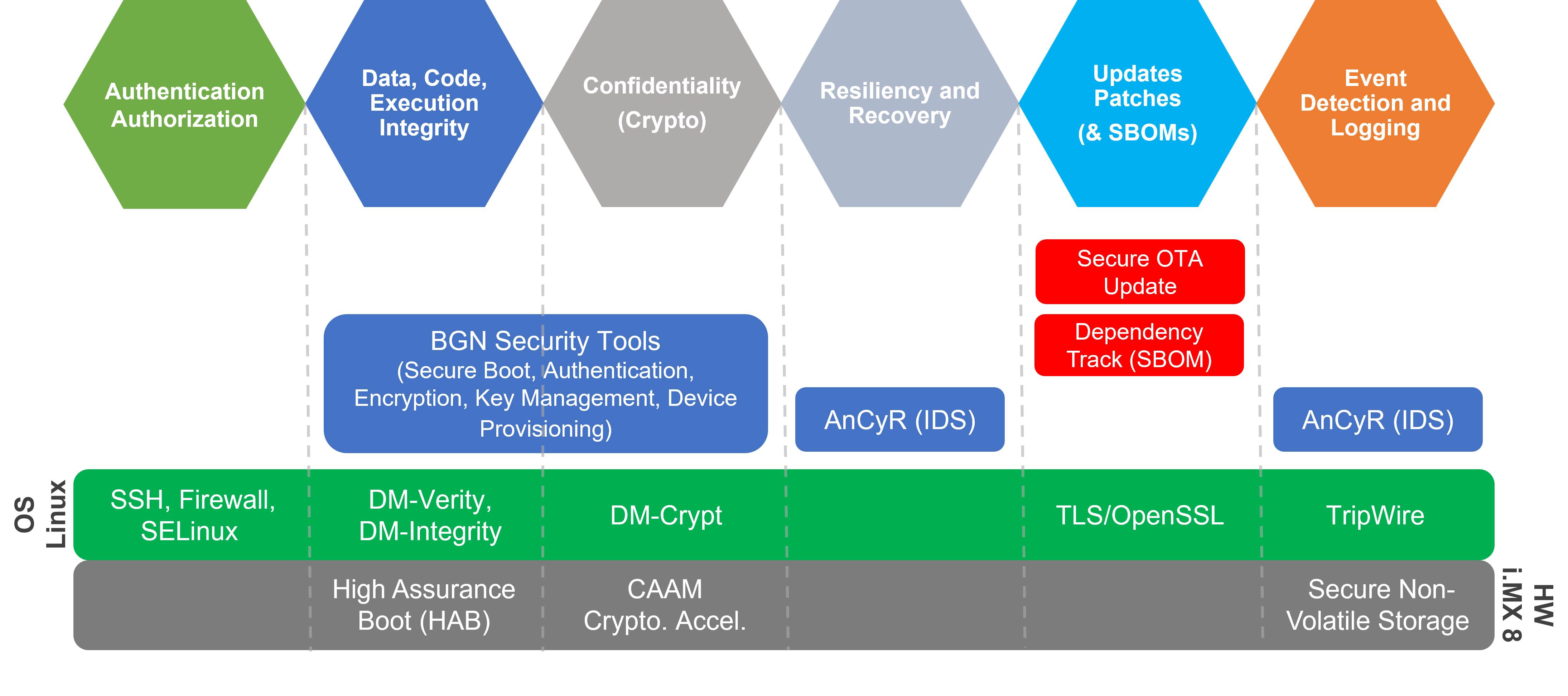
BG Networks Cybersecurity Solution Map
BG Networks offers security automation tools that saves you significant time to meet FDA device cybersecurity requirements. All 6 categories of device security, specified by the FDA, are covered with BG Networks automation solutions.